Serum osmolality10/16/2023 
Otherwise, one litre of plasma would be equivalent to one kilogram of plasma, and plasma osmolarity and plasma osmolality would be equal. 
For a given solution, osmolarity is slightly less than osmolality, because the total solvent weight (the divisor used for osmolality) excludes the weight of any solutes, whereas the total solution volume (used for osmolarity) includes solute content. In contrast, osmolality is independent of temperature and pressure. Osmolarity is affected by changes in water content, as well as temperature and pressure. 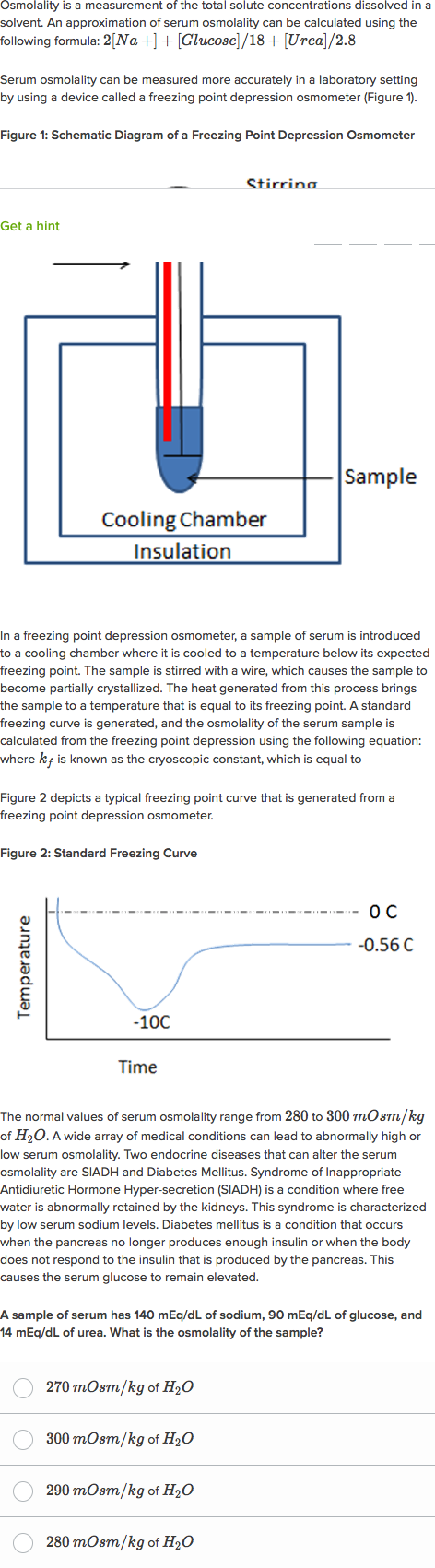
It works on the method of depression of freezing point. Osmolality can be measured on an analytical instrument called an osmometer.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
